Post by DirtDiva Admin on Aug 18, 2020 10:33:24 GMT -6
This came with my latest oxygen absorber purchase thought it might be useful info. From PackFreshUSA
The Science Of Oxygen Absorbers
Oxygen Absorbing Packets (Oxygen Absorbers) are rated according to how much oxygen they will absorb, e.g., a 100cc Oxygen Absorbers will absorb 100cc's of oxygen. There are different types of oxygen absorbers, but PackFreshUSA only sells the most versatile type. They contain an iron carbonate formulation, along with a tiny amount of entrapped water, and they chemically trap available oxygen from the environment. This process creates iron oxide, which is the same process that makes a nail rust. This reaction is exothermic, meaning that heat is released as the oxygen is absorbed, but not enough to be of concern during normal use and you likely will not even notice.
Oxygen absorbers are not air absorbers. Sometimes users expect to see a dramatic vacuum effect, but while it is true that you are likely to notice a volume decrease, oxygen is only 21% of air and the rest of the air will always remain, regardless of how many Oxygen Absorbers are used. The rest of the air is mostly nitrogen, with trace amounts of some other gases that are inconsequential to food storage.
Calculations For Oxygen Absorbers
First, we need to know the volume of the container we are going to deoxygenate in cc's. (cubic centimeters) Oxygen, as a gas, is measured in cc's, and containers are typically measured in milliliters, (ml) but conveniently, 1 ml = 1 cc.
Google is great for this: For example, if we have a pint jar, you can type in the search bar, “1 pint in mls” or “16oz in mls” and you will learn that a pint jar is roughly 473ml which is 473cc's. So, a pint jar holds 473 cc's of air.
Now we need to calculate how much oxygen is in the jar. (To make the calculations easier, we will use 20% or 1/5 as the oxygen content of air) Because oxygen is 20% or 1/5th or air, we can divide:
473cc/5 = 94.6 cc
So, an EMPTY pint jar contains 94.6 cc's of oxygen and will be deoxygenated by using an Oxygen Absorbers of 100cc's or larger. You can also fill that jar with product and still use a 100cc Oxygen Absorbers, OR you could account for the volume of the product and use a smaller Oxygen Absorbers...
To account for the product volume, you need to know how much air is in the product. Air is contained in the product in two ways: embedded air contained within the food particles and intersial air in-between the food particles. There can be a surprising amount of air within the food itself and this air needs to be accounted for.
A lot of misinformation on the internet is related to people thinking items with small particles (like flour) are “dense” and therefore have less air volume, but particle size is actually not important. Believe it or not, a pool filled with marbles will have the same intersial air volume as one filled with basketballs. Yes, the basketballs will have much larger pockets of air, but there are far fewer of them. Think of a checkerboard with just 2 large black squares and 2 large white squares... ½ of it's area is black and ½ white. What if it had 100 small black squares and 100 small white ones? 1000? Even with tiny squares it is still ½ black and ½ white as long as the squares are all a uniform size.
What does matter is particle size distribution, or in other words, whether you have particles that are all a uniform size or if you have a lot of particles of different sizes. If you have a pool filled with basketballs AND marbles, the marbles can fill in all the large pockets of air. So, products with uniform particle sizes will have more intersial air than products with a greater distribution of particle sizes.
The other really important factor is degree of agglomeration, which just means how tightly everything is packed. Unsifted flour contains less air than sifted flour because it is more agglomerated. It is always best to pack food tightly to give it the smallest possible air volume.
How can you possibly know the particle size distribution and degree of agglomeration of your food products? There is a very simple test you can perform that will give you a very good idea of the air volume: If you take 2 cups of water and 1 cup of your product, then mix them really good and measure the result, you will end up with something less than 3 cups. The difference between 3 cups and the measured volume of your “product soup” is the amount of air that was in that cup of product. So,
Air volume of 1 cup of product = 3 cups – (volume of product soup in cups)
If your product soup measures 2 ½ cups, you know that ½ cup of the product was air, or it was 50% air by volume. If your product soup measures 2 ¼ cups, then ¾ cup of the product was air, or it was 75% air by volume.
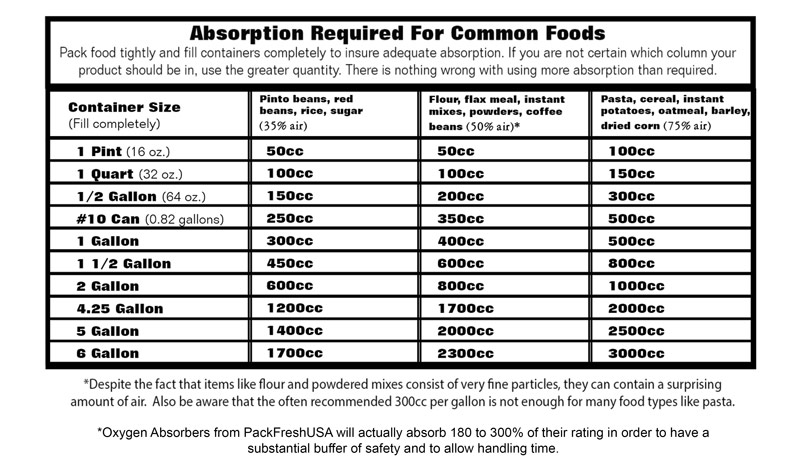
The following chart shows the air volumes of some popular LTFS products and the absorption required by container size:
The Science Of Oxygen Absorbers
Oxygen Absorbing Packets (Oxygen Absorbers) are rated according to how much oxygen they will absorb, e.g., a 100cc Oxygen Absorbers will absorb 100cc's of oxygen. There are different types of oxygen absorbers, but PackFreshUSA only sells the most versatile type. They contain an iron carbonate formulation, along with a tiny amount of entrapped water, and they chemically trap available oxygen from the environment. This process creates iron oxide, which is the same process that makes a nail rust. This reaction is exothermic, meaning that heat is released as the oxygen is absorbed, but not enough to be of concern during normal use and you likely will not even notice.
Oxygen absorbers are not air absorbers. Sometimes users expect to see a dramatic vacuum effect, but while it is true that you are likely to notice a volume decrease, oxygen is only 21% of air and the rest of the air will always remain, regardless of how many Oxygen Absorbers are used. The rest of the air is mostly nitrogen, with trace amounts of some other gases that are inconsequential to food storage.
Calculations For Oxygen Absorbers
First, we need to know the volume of the container we are going to deoxygenate in cc's. (cubic centimeters) Oxygen, as a gas, is measured in cc's, and containers are typically measured in milliliters, (ml) but conveniently, 1 ml = 1 cc.
Google is great for this: For example, if we have a pint jar, you can type in the search bar, “1 pint in mls” or “16oz in mls” and you will learn that a pint jar is roughly 473ml which is 473cc's. So, a pint jar holds 473 cc's of air.
Now we need to calculate how much oxygen is in the jar. (To make the calculations easier, we will use 20% or 1/5 as the oxygen content of air) Because oxygen is 20% or 1/5th or air, we can divide:
473cc/5 = 94.6 cc
So, an EMPTY pint jar contains 94.6 cc's of oxygen and will be deoxygenated by using an Oxygen Absorbers of 100cc's or larger. You can also fill that jar with product and still use a 100cc Oxygen Absorbers, OR you could account for the volume of the product and use a smaller Oxygen Absorbers...
To account for the product volume, you need to know how much air is in the product. Air is contained in the product in two ways: embedded air contained within the food particles and intersial air in-between the food particles. There can be a surprising amount of air within the food itself and this air needs to be accounted for.
A lot of misinformation on the internet is related to people thinking items with small particles (like flour) are “dense” and therefore have less air volume, but particle size is actually not important. Believe it or not, a pool filled with marbles will have the same intersial air volume as one filled with basketballs. Yes, the basketballs will have much larger pockets of air, but there are far fewer of them. Think of a checkerboard with just 2 large black squares and 2 large white squares... ½ of it's area is black and ½ white. What if it had 100 small black squares and 100 small white ones? 1000? Even with tiny squares it is still ½ black and ½ white as long as the squares are all a uniform size.
What does matter is particle size distribution, or in other words, whether you have particles that are all a uniform size or if you have a lot of particles of different sizes. If you have a pool filled with basketballs AND marbles, the marbles can fill in all the large pockets of air. So, products with uniform particle sizes will have more intersial air than products with a greater distribution of particle sizes.
The other really important factor is degree of agglomeration, which just means how tightly everything is packed. Unsifted flour contains less air than sifted flour because it is more agglomerated. It is always best to pack food tightly to give it the smallest possible air volume.
How can you possibly know the particle size distribution and degree of agglomeration of your food products? There is a very simple test you can perform that will give you a very good idea of the air volume: If you take 2 cups of water and 1 cup of your product, then mix them really good and measure the result, you will end up with something less than 3 cups. The difference between 3 cups and the measured volume of your “product soup” is the amount of air that was in that cup of product. So,
Air volume of 1 cup of product = 3 cups – (volume of product soup in cups)
If your product soup measures 2 ½ cups, you know that ½ cup of the product was air, or it was 50% air by volume. If your product soup measures 2 ¼ cups, then ¾ cup of the product was air, or it was 75% air by volume.
The following chart shows the air volumes of some popular LTFS products and the absorption required by container size:


